Abt. Enzymologie - Postranslationale Modifikationen von Proteinen


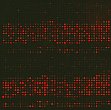
Die Interaktion von Proteinen mit ihren Bindungspartnern oder Substraten wird durch eine Vielzahl verschiedener posttranslationaler Modifikationen reguliert. Moderne massenspektrometrische Methoden haben zu einem explosionsartigen Zuwachs an bekannten Modifikationsstellen in den verschiedenen Proteomen geführt. Allerdings ist bei dieser beschreibenden Analyse oft nicht klar, welche Enzyme diese Modifikationen einbauen und wie die Wechselwirkung zwischen den verschiedenen posttranslationalen Modifikationen sind. Unsere Arbeitsgruppe entwickelte die SPACE-CHIP Technologie (Singel Peptide Arrays for Characterization of Enzymes), um enzymatische Reaktionen auf Peptid-Mikroarrays systematisch untersuchen zu können. Schwerpunkte der Abteilung bilden Untersuchungen:
- zu den molekularen Mechanismen proteinmodifizierender Enzyme unter besonderer Berücksichtigung von Kinasen, Acetyltransferasen, Methyltransferasen und Histondeacetylasen
- zur Entwicklung kontinuierlicher Enzymassays, besonders für Enzyme, die posttranslationale Modifikationen aus Proteinen entfernen können
- zur Spezifität von Proteindomänen, die posttranslational modifizierte Proteine erkennen





